ELECTRONIC SPHYGMOMANOMETER, manual inflation(Microlife VSA)
Valid Article
ELECTRONIC SPHYGMOMANOMETER, Microlife VSA
Definition
Handheld semi-automatic oscillometric device to measure blood pressure in people aged 12 years or older.
An electronic device, typically digital, designed to noninvasively measure blood pressure using a manually-inflated cuff that fits around the arm. It displays current heart rate in addition to systolic and diastolic blood pressures; it has memory to store blood pressure values and a visual warning system if blood pressure exceeds pre-set limits. This device is designed to be portable (e.g., hand-held or carried in a pouch).
Synonym
Vital Signs Alert - CRADLE device
Vital Signs Monitor (Cradle) for pregnant women
Specifications
The device incorporates a traffic light early warning system, aiming to alert in a visual way (color code) to abnormalities in vital signs secondary to pre-eclampsia or shock (due to bleeding or sepsis). It is suitable for use in low-resource settings according to WHO criteria.
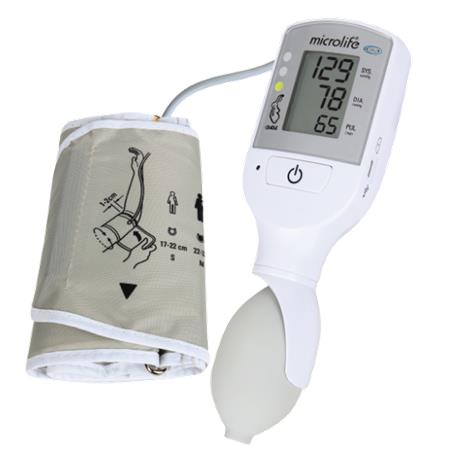
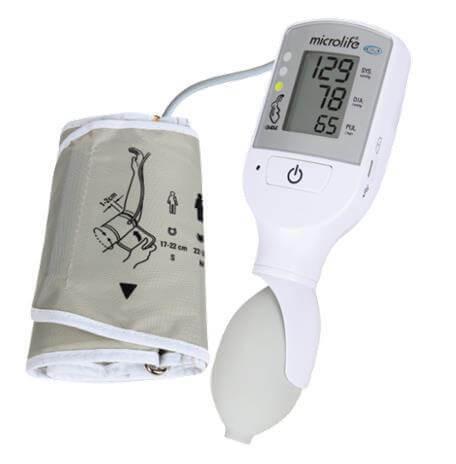
- measuring unit:
- ON/OFF button
- LCD-display
- cuff socket
- battery compartment
- Micro USB port (for charging only)
- traffic light
- charging indicators
- cuff: the VSA device comes with a medium cuff as standard
- cuff connector
- pump ball: for manual inflation of the cuff
Technical specifications
- Weight: 157.7 g (including battery)
- Dimension: 167 x 60 x 41 mm
- Display: LCD-Display (Liquid Crystal Display)
- Measuring procedure: oscillometric, corresponding to Korotkoff method Phase I systolic, Phase V diastolic.
- measurement range: 30-280 mmHg blood pressure
- 40-200 beats per minute-pulse
- cuff pressure display range: 0-299 mmHg
- Resolution: 1 mmHg
- Static accuracy: pressure within ± 3 mmHg
- Pulse accuracy: ± 5% of the readout value
- Voltage source: specify battery type (3.7V, 350 mAh), rechargeable via standard MicroUSB
- Battery life: approx. 200 measurements per recharge cycle
Supplied with the Article
- Cuff size M for circumference of upper arm 22 - 32 cm
- Battery type 3.7V, 350 mAh, rechargeable via standard MicroUSB
- USB cable
To be Ordered Separately
Cuff size L for circumference of upper arm 32 - 42 cm
Instructions for use
Read carefully the user manual
Maintenance
- Device care: Clean the device only with a soft, dry cloth
- Cleaning the cuff: take out the bladder. Clean the cuff with warm water and soap detergent. Air dry.
Storage
Do not expose the device to either extreme temperatures,
humidity, dust or direct sunlight.
- Storage conditions: -20 - +55 °C; 15 - 90 % relative maximum humidity
- Operation conditions: 10 - 40 °C; 15 - 90 % relative maximum humidity
MSF requirements
Meets the World Health Organisation (WHO) criteria for devices suitable for in low-resource settings
It can be used with USB phone chargers.
Requires minimal training. It is meant for monitoring antenatal blood pressure screening in pregnant women in remote areas by health community workers or trained health workers to facilitate the decision of referring the pregnant women to a hospital structure.

![[EMEQSPHY5CL] (sphygmomanometer Microlife) CUFF adult L 32-42cm](/web/image/product.template/577974/image_256/%5BEMEQSPHY5CL%5D%20%28sphygmomanometer%20Microlife%29%20CUFF%20adult%20L%2032-42cm?unique=5602501)