CLAVICLE FRACTURE BRACE, size L
STD
EPHYCLFB1L-
Valid Article
Account code:
60210
HS Code:
901910
Last Updated on:
13/11/2025, 23:17:26
Former
Code(s):
EPHYZFR0083
CE marking: declaration that the product meets EU standards for health, safety, and environmental protection. The CE marking indicates that the product may be sold freely in any part of the European Economic Area, regardless of its country of origin.
M030501 - Immobilisation bandages for shoulder, arm and collarbone
European Medical Device Nomenclature (EMDN) is the nomenclature of use by manufacturers when registering their medical devices in the EUDAMED database. EMDN is characterised by its alphanumeric structure that is established in a seven-level hierarchical tree.
In Europe, medical material that fulfills the definition of a medical device according to the MDR (Medical Device Regulation) is classified into 4 classes
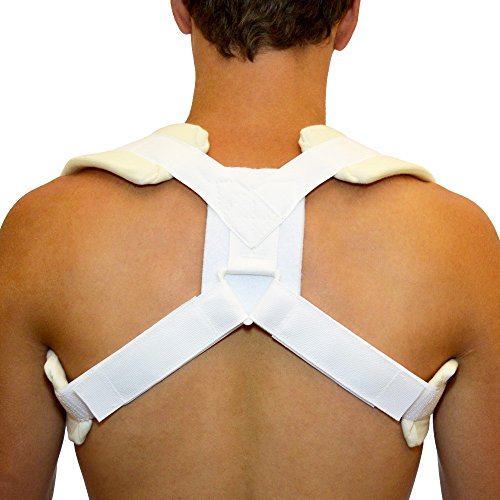
CLAVICLE FRACTURE BRACE
Definition
A device intended to be worn over the shoulders of a patient to provide support and protection to a clavicle bone which has been fractured or injured. It is formed as a figure-of-eight harness that descends from the shoulders, passes under each arm and crosses over the back. It has broad, padded straps and may have extra padding inserted at the back. This device may also be used to improve the patient's posture. This is a reusable device.
Synonym
8 splint,clavicular splint
Specifications
- harness as a figure of 8 with broad padded straps
- 3 sizes: small, medium and large
Instructions for use
The shoulders have to be pulled backwards until a perfect alignment of the broken clavicle.
Some restricted information has been hidden. Sign in
to see this information
Maintenance
Follow the manufacturer's indications for cleaning.
Some restricted information has been hidden. Sign in
to see this information