(mb HAIN LPA) TEST, GENOTYPE MTBDRplus v2.0, 1 test, 30496A
Valid Article
(mb HAIN LPA) TEST, GENOTYPE MTBDRplus, v2.0
Reserved for TB culture programmes.
CAUTION
This test is worded and codified by unit to make the order easier, but it is always packaged in kit of 96.
Definition
Line probe assays (LPA) are a family of DNA strip-based tests that allow users to determine the drug resistance profile of an MTB strain by interpreting a pattern of bands that represent lines of immobilized probes that are bound (or hybridized) to MTB amplicons (DNA amplification products). LPA probes are designed to target the most common mutations associated with resistance to first- and second-line anti-TB agents and specific MTBC wild-type (WT) DNA sequences.
The GenoType MTBDRplus VER 2.0 is a qualitative in vitro test for the identification of the Mycobacterium tuberculosis complex and its resistance to rifampicin (RMP) and isoniazid (INH) from pulmonary smear-positive or -negative clinical specimens and cultivated samples.
The technique is based on a combination of PCR and DNA strip technology.
Specifications
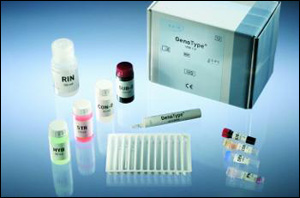
Components
KIT composed of 2 separate elements with different storage conditions:
- component 1/2 (cardboard box) to be stored between 2° and 8°C
- component 2/2 (small bag) to be stored at -20°C (deep freeze)
Technical specifications
Kit contains sufficient reagents for analysis of 96 samples.
Kit Component 1 of 2 (store at 2-8°C)
- Membrane strips coated with specific probes (MTBDRplus VER 2.0 STRIPS), 2x48 strips
- Denaturation Solution (DEN) contains <2% NaOH, dye, 2x1.2 ml
- Hybridization Buffer (HYB) contains 8-10% anionic tenside, dye, 120 ml
- Stringent Wash Solution (STR) contains >25% of a quaternary ammonium compound, <1% anionic tenside, dye, 120 ml
- Rinse Solution (RIN) contains buffer, <1% NaCl, <1% anionic tenside, 3x120 ml
- Conjugate Concentrate (CON-C) contains streptavidin-conjugated alkaline phosphatase, dye, 1.2 ml
- Conjugate Buffer (CON-D) contains buffer, 1% blocking reagent, <1% NaCl, 120 ml
- Substrate Concentrate (SUB-C) contains dimethyl sulfoxide, substrate solution, 1.2 ml
- Substrate Buffer (SUB-D) contains buffer, <1% MgCl2, <1% NaCl, 120 ml
- tray, evaluation sheet, 4 of each
- instructions for use, template, 1 of each
Kit Component 2 of 2 (store at –20°C)
- Amplification Mix A (AM-A GT MTBDRplus VER 2.0) contains buffer, nucleotides, Taq polymerase, 4x 0.3 ml
- Amplification Mix B (AM-B GT MTBDRplus VER 2.0) contains salts, specific primers, dye, 4x 1.05 ml
Quality control
In order to control the correct performance of the test and the proper functioning of kit constituents, each strip includes 5 control zones:
- a Conjugate Control zone (CC) to check the binding of the conjugate on the strip and a correct chromogenic reaction
- an Amplification Control zone (AC) to check for a successful amplification reaction
- three Locus Control zones (rpoB, katG, and inhA) checking the optimal sensitivity of the reaction for each of the tested gene loci
A negative control sample for detection of possible contamination events containing water (molecular biology grade) instead of DNA should be part of each test run; the respective test strip should show the bands CC and AC only.
Packaging & Labelling
1 kit = 96 tests
To be Ordered Separately
Material Required but not Included in the Kit: see checklist molecular biology Hain.
Instructions for use
The GenoType MTBDRplus test is based on the DNA STRIP technology. The whole procedure is divided into three steps:
- DNA extraction from clinical specimens (pulmonary, decontaminated) or cultured material (solid/liquid medium) – the necessary reagents are not included in the kit
- a multiplex amplification with biotinylated primers
- a reverse hybridization.
Visual interpretation of the results.
Precautions for Use
- It is essential that all materials (such as pipette tips) coming in contact with the reagents are free from DNases.
- Do not interchange or pool Amplification Mixes or membrane strips from different kits unless the lots are identical.
- When handling kit reagents, the following special safety measures must be applied:
- The denaturation Solution (DEN) contains <2% NaOH and is irritating to eyes and skin (R36/38 and S26-37/39-45).
- The Substrate Concentrate (SUB-C) contains dimethyl sulfoxide and is irritating (R36/37/38, S23-26-36).
Storage
- Store all constituents from Kit Component 1 between 2° and 8°C.
- Store all constituents from Kit Component 2 at –20°C and keep strictly separated from contaminating DNA.
- Avoid repeated freezing and thawing of AM-A and AM-B; when processing only small sample numbers per run, aliquot AM-A and AM-B.
- Shelf life: 12 to 15 months
- Guaranteed minimum remaining shelf life at delivery: 1/3 of total shelf life
- Do not use the reagents beyond their expiry date.
Waste management
- Dispose of unused reagents and waste in accordance with federal, state, and local regulations.
- Discard used pipette tips immediately after use in a container for biohazardous waste.
- Discard all used disposables in a container
for biohazardous waste to be incinerated.
MSF requirements
Drug susceptibility testing
Molecular testing requires specific laboratory set up, equipment and trained personnel. Refer to your lab advisor.

![[ELAEMBIE51-] INCUBATOR (TwinCubator), 4 - 99°C, 12 samples](/web/image/product.template/570762/image_256/%5BELAEMBIE51-%5D%20INCUBATOR%20%28TwinCubator%29%2C%204%20-%2099%C2%B0C%2C%2012%20samples?unique=a61b873)
![[ELAEMBIE52-] THERMAL CYCLER (GTQ-Cycler 96), 96 samples](/web/image/product.template/570761/image_256/%5BELAEMBIE52-%5D%20THERMAL%20CYCLER%20%28GTQ-Cycler%2096%29%2C%2096%20samples?unique=a61b873)