ROOT ELEVATOR, APICAL, n°2, 325-13-02
Valid Article
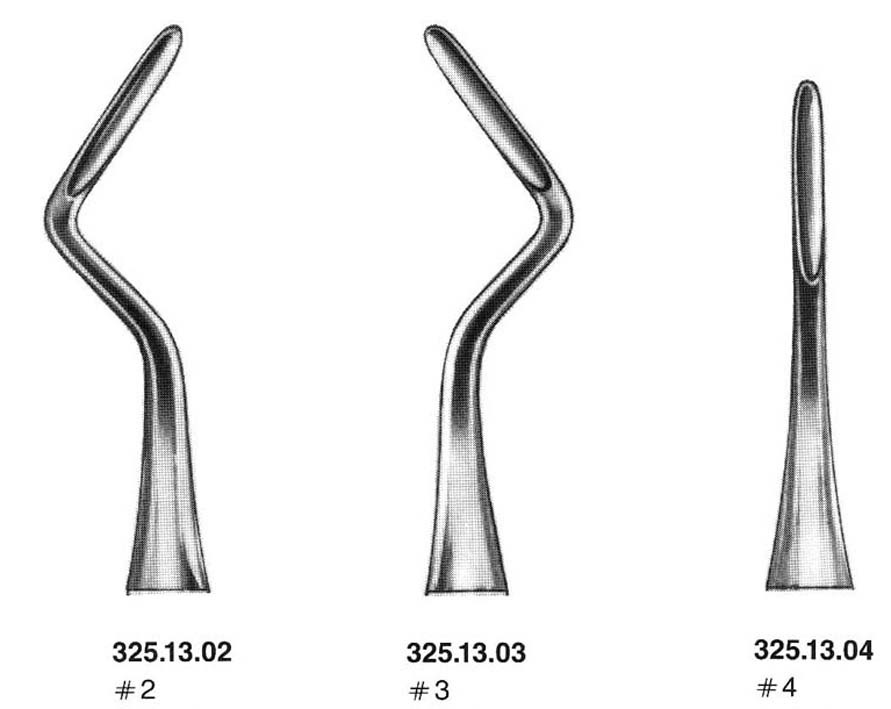
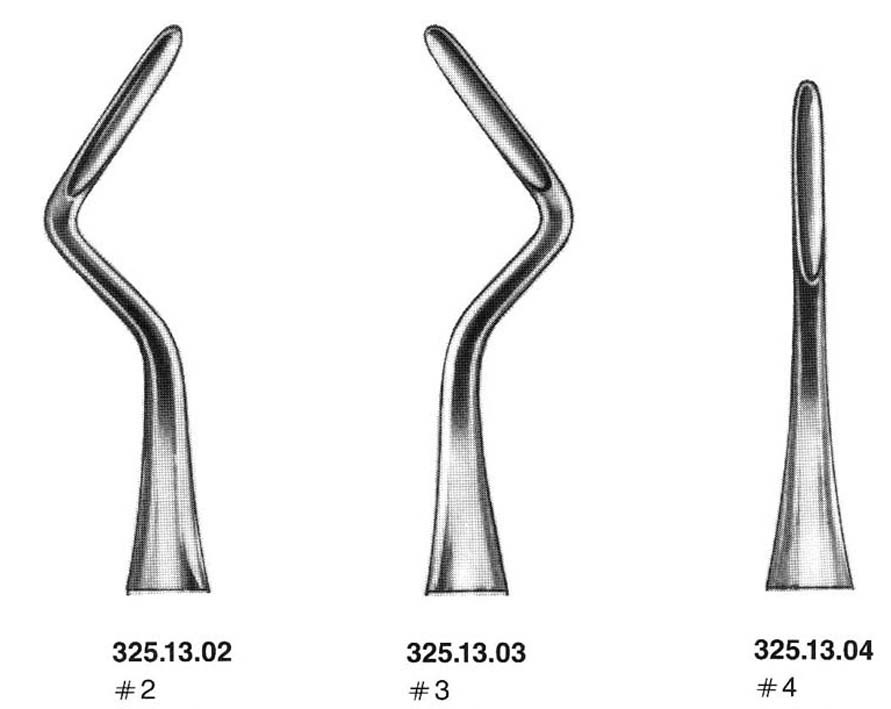
ROOT ELEVATOR, APICAL
Definition
A hand-held dental instrument used as a lever during the extraction of a tooth or retained roots. It is a one-piece instrument with a strong but slender blade at the distal working end. It is pushed in between the gums and the tooth exterior to loosen the tissue surrounding the tooth. Sometimes this can be enough to loosen and extract the tooth without the use of other instruments.
Specifications
3 different shapes:
- curved nº 2: left side
- curved nº 3: right side
- straight nº 4
Instructions for use
The instruments are delivered “non-sterile”.
They mustbe used by trained, qualified personnel who respect the recommendations given in the instructions for use.
Maintenance
General recommendations
- It is recommended to reprocess instruments as soon as possible after use
- Prohibit the use of sodium hypochlorite to avoid corrosion pitting on instruments
- For all metal appliances, it is recommended to use anti-corrosion cleaning and disinfection agents.
- Do not use fixing products or hot water (> 40°C) which will cause the proteins to fix and influence the cleaning result.
- Follow the instructions and respect the concentrations and immersion times given by the manufacturer (excessive concentration can lead to corrosion or other defects on the devices)
- Perform a visual inspection to verify the cleanliness and integrity of the instruments. If necessary, repeat the reprocessing process until the instrument is visibly clean.
- It is very important to check all surgical instruments before use for visible wear or damage.
Due to the design of the product and the materials used, no limits on the maximum number of processing cycles achievable can be specified. The lifespan of medical devices is determined by their function and careful handling.

![[KSUDBDEN20-] DENTAL SET, 20 instruments](/web/image/product.template/569814/image_256/%5BKSUDBDEN20-%5D%20DENTAL%20SET%2C%2020%20instruments?unique=4528210)